
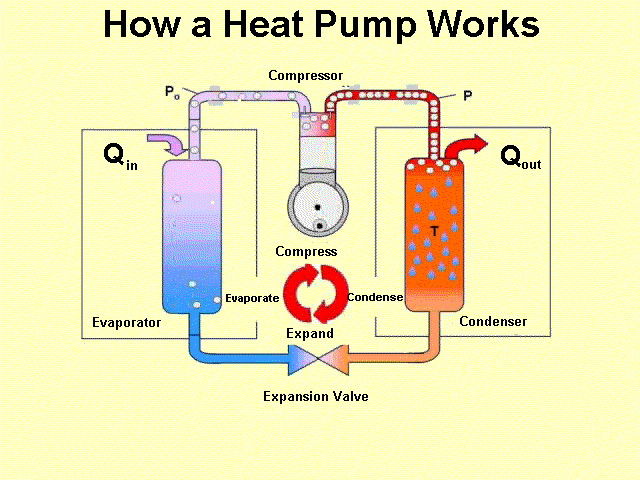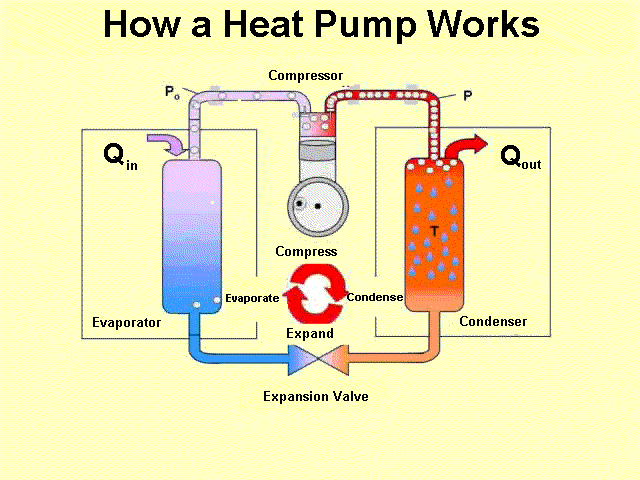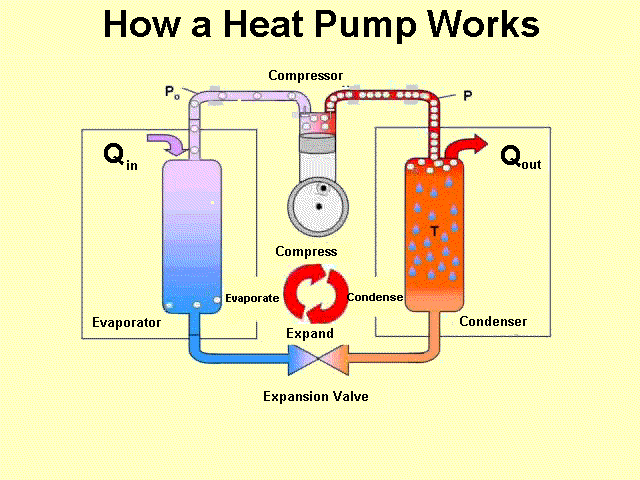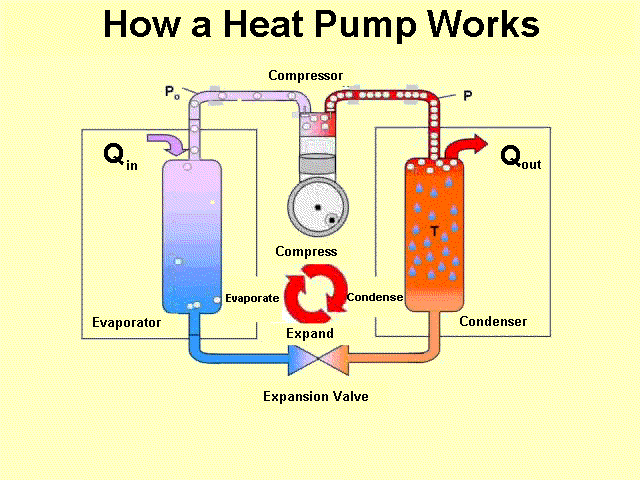
Heat
Proficiency Standards
B03-Matter

B04-Chemistry
 |
 Heat |
 Proficiency Standards |
B03-Matter |
 B04-Chemistry |
| B02 Atoms |
| What are atoms? |
 What's different between atoms? What's different between atoms? (Mastery) |
 Which atoms are important for me? Which atoms are important for me? (Interpersonal) |
 How do we categorize atoms? How do we categorize atoms?(Understanding) |
 How are atoms spread through the universe? How are atoms spread through the universe?(Self Expressive) |
|
Practice
Quiz: SCA_B02 Atoms Take every day before sleeping! Quia Atoms Math Practice Vocabulary Review Activities BrainPop Animations and Practice Quizes 
|
Vocabulary Atoms: Chapter 2 Section 1: Pages 50-54: The Periodic Table: Chapter 2 Section 2: Pages 55-58 Atoms Reader Elements Reader Matter Everywhere Booklet Science Skills Handbook Appendix: Pages 202-214: Process Skills Packet (Online Textbook: Log onto Pearson.com, then click on the titles above for the online text.) |
| Labs & Videos |
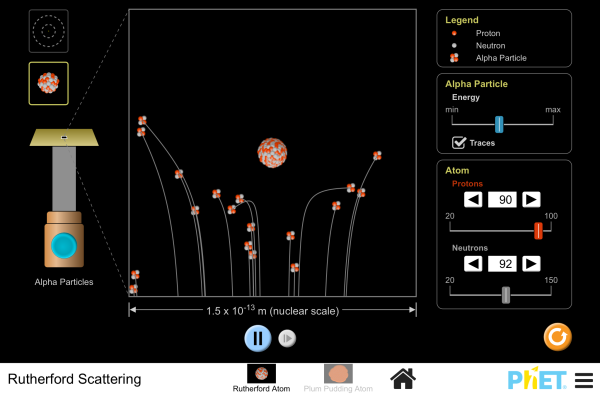 Rutherford Scattering write up an experiment based on this activity. |
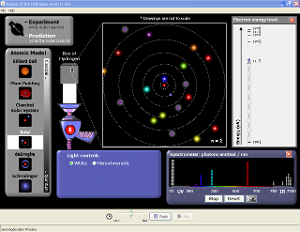 Hydrogen Atom write up an experiment based on this activity. |
 Build an Atom write up an experiment based on this activity. |
 Microwaves write up an experiment based on this activity. |
 Alpha Decay write up an experiment based on this activity. |
 Beta Decay write up an experiment based on this activity. |
 Isotopes and Atomic Mass write up an experiment based on this activity. |
 Atomic Interactions write up an experiment based on this activity. |
 Gizmos Atom Builder Worksheet |
 Gizmos Nuclear Decay Worksheet |
 Gizmos Bohr Model Worksheet |
 Gizmos Electron Configuration Worksheet |
 Nuclear Fission |
Build
an Atom PowerPoint Interactive |
 Hands-On CERN |
Chem4Kids.com |
 Fermilab Games |
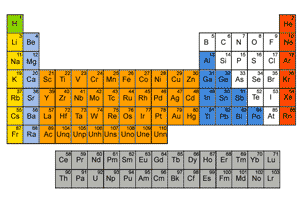 Interactive Periodic Table |
 Jefferson Lab Atom Tour Activity |
 Visual Periodic Table |
Pearson Videos
|

|
Fermilab Videos Detecting Particles Accelerating Particles
|
|
 |
 |
||
 |
 |

|
 |
| Engage Discrepant Event |
Explore Research |
Explain Write-Up |
Elaborate New situations/applications |
Evaluate project to share |
| Reading & Math Work |
|
|
|
| Projects by Learning Style and Media Type |
 Sensing-Thinking
(Mastery) Sensing-Thinking
(Mastery)Facts
|
 Sensing-Feeling
(Interpersonal) Sensing-Feeling
(Interpersonal)A time when you...
|
 Intuitive-Thinking
(Understanding) Intuitive-Thinking
(Understanding)Playing with facts
|
 Intuitive-Feeling
(Self-Expressive) Intuitive-Feeling
(Self-Expressive)Creating new possibiliteis
|
|
|
|
 Live
Presentation Project Live
Presentation Project
|
| Essential Vocabulary & Concepts |
| Picture | Core Knowledge or
Concept |
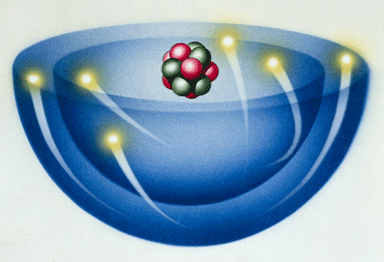 |
An
atom is the smallest
particle of a substance (matter). |
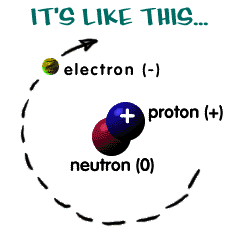 |
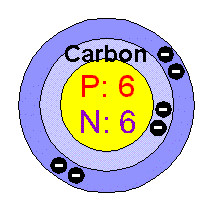
There are 2 regions of an atom The nucleus in the tiny center of the atom is made of protons and neutrons. The electron cloud is the outside area made up of mostly empty space. |
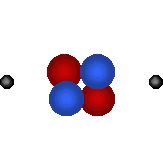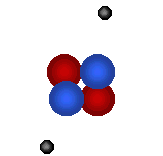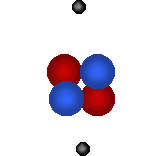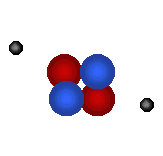 |
Protons
are tiny positively charged
particles. Neutrons are neutral (no charge). |
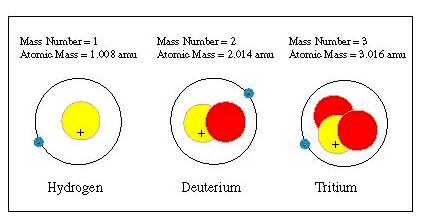 |
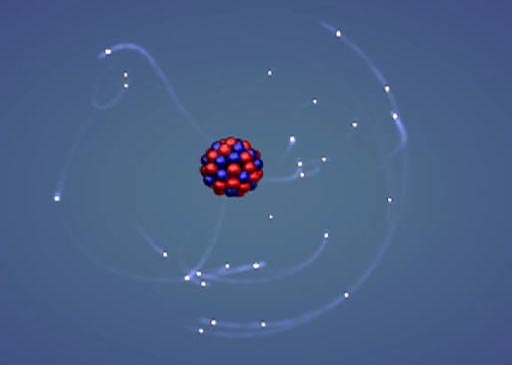
Atomic mass
is the total number of an atom’s protons and neutrons. Atomic number is the number of an atom’s protons only. This number is used to classify types of atoms. |
 |
Electrons
are negatively charged
particles that orbit around the nucleus in the
electron cloud. They are much smaller than protons and neutrons. In a neutral atom, there are the same number of electrons as protons. Valence electrons react with other atoms. Thry have the most energy and are the furthest from the nucleus. |
 |
A pure substance is stuff made of only one kind of matter with definite properties. (i.e. sugar, salt, iron, & copper) |
 |
Some pure substances, called elements, cannot be broken down into other substances by any chemical means. (i.e. iron, copper, gold, carbon, & chlorine) |
 |
The periodic table an arrangement of the chemical elements by increasing atomic number, which displays the elements so that one may see trends in their properties. |
 |
Elements in the same group (column)
react similarly. Group1 and 2 metals react strongly with group 7 elements. Group 8 gasses are unreactive. |
 |
Scientist MarieCurie (ne: Sklodowska): the first person to receive 2 Nobel Prizes for her work in radioactive particle physics (1903) and chemistry (1911). Her work paved the way for modern nuclear technologies in medicine, power, and the military. |
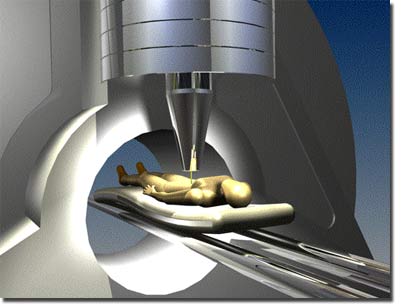 |
Technology Proton Therapy uses a beam of massive protons to deliver more energy to destroy cancer cells with less danger to the patient. |